Bond Length Chart
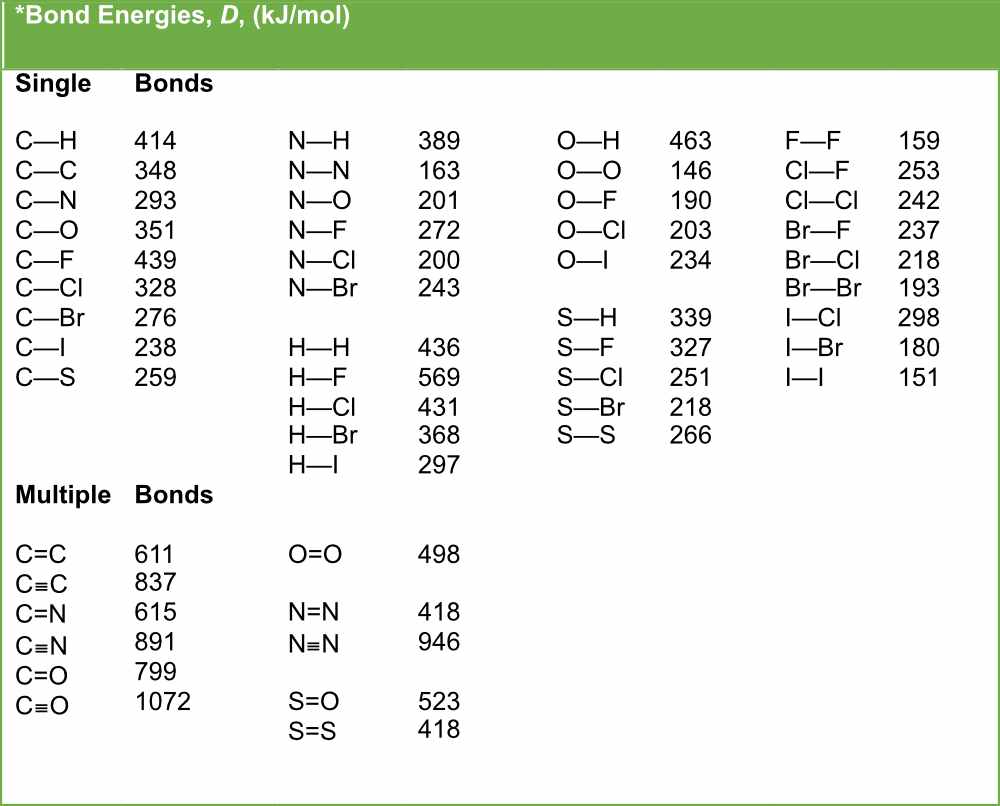
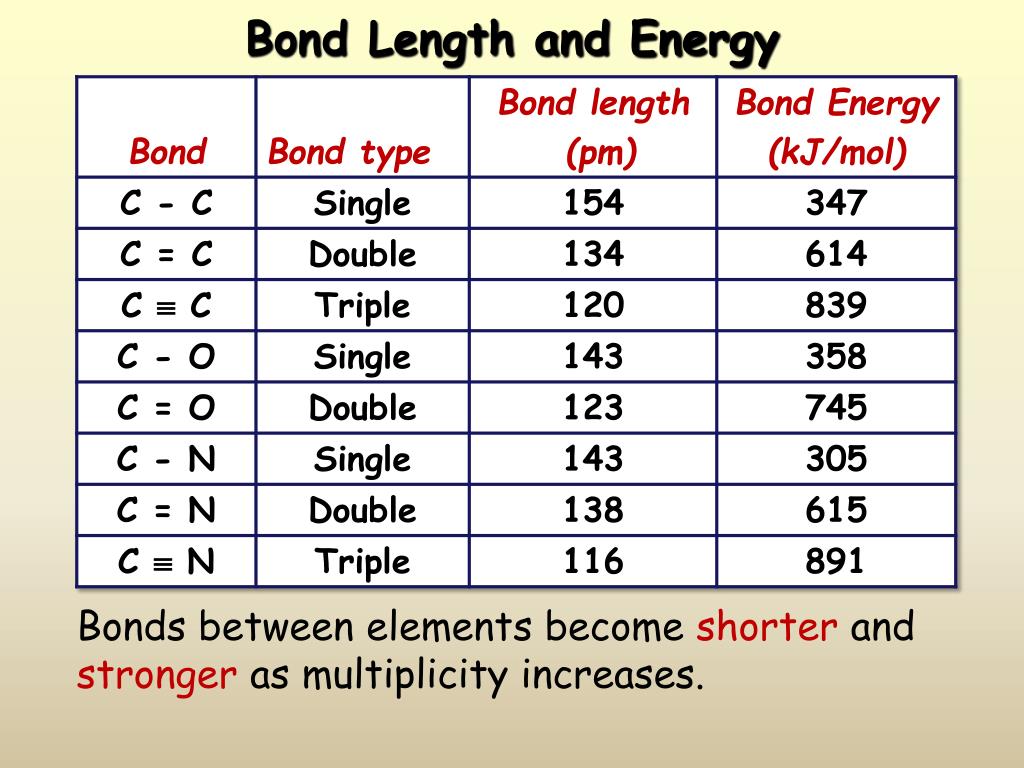
Bond Length Chart - The length of the bond is determined by the number of bonded electrons (the bond order). Web below is a table of average bond lengths. Web in molecular geometry, bond length or bond distance is defined as the average distance between nuclei of two bonded atoms in a molecule. Web the amount of energy required to break all covalent bonds of the same type in one mole of a compound in a gaseous state is called bond energy. Bond order is the number of electron pairs that hold two atoms together. Lewis diagram of formaldehyde (ch₂o) It is a transferable property of a bond between atoms of fixed types, relatively independent of the rest of the molecule. This is because as we go down the group, the atomic radius increases. Before we go into the details explaining the bong lengths and bond strengths in organic chemistry, let’s put a small summary for these two properties right from the beginning as it stays relevant for all types of bonds we are going to talk about. Bond energies and bond lengths. Lewis diagram of formaldehyde (ch₂o) Bond length and bond strength. As we go across a period we see bond length decreases. Bond energies and bond lengths. A bond is not static but dynamic with the atoms undergoing the attractive and repulsive forces as described in the potential well (fig. Single bonds have a bond order of one, and multiple bonds with bond orders of two (a double bond) and three (a triple bond). The higher the bond order, the stronger the pull between the two atoms and the shorter the bond length. Single and multiple covalent bonds. Before we go into the details explaining the bong lengths and bond strengths in organic chemistry, let’s put a small summary for these two properties right from the beginning as it stays relevant for all types of bonds we are going to talk about. Web the amount of energy required to break all covalent bonds of the same type in one mole of a compound in a gaseous state is called bond energy. Web in molecular geometry, bond length or bond distance is defined as the average distance between nuclei of two bonded atoms in a molecule. Web the amount of energy required to break all covalent bonds of the same type in one mole of a compound in a gaseous state is called bond energy. Web bond length is defined as the. Bond order is the number of electron pairs that hold two atoms together. A bond is not static but dynamic with the atoms undergoing the attractive and repulsive forces as described in the potential well (fig. Web from this graph, we can determine the equilibrium bond length (the internuclear distance at the potential energy minimum) and the bond energy (the. The higher the bond order, the stronger the pull between the two atoms and the shorter the bond length. As we go across a period we see bond length decreases. Bond length and bond strength. 8.9.1), where the bottom of the well represents the equilibrium position of the oscillating atoms, which. It is a transferable property of a bond between. Browse videos, articles, and exercises by topic. A bond is not static but dynamic with the atoms undergoing the attractive and repulsive forces as described in the potential well (fig. Web in molecular geometry, bond length or bond distance is defined as the average distance between nuclei of two bonded atoms in a molecule. The length of the bond is. Single bonds have a bond order of one, and multiple bonds with bond orders of two (a double bond) and three (a triple bond). Bond length and bond strength. Web from this graph, we can determine the equilibrium bond length (the internuclear distance at the potential energy minimum) and the bond energy (the energy required to separate the two atoms).. Web the amount of energy required to break all covalent bonds of the same type in one mole of a compound in a gaseous state is called bond energy. Before we go into the details explaining the bong lengths and bond strengths in organic chemistry, let’s put a small summary for these two properties right from the beginning as it. Single and multiple covalent bonds. Web the amount of energy required to break all covalent bonds of the same type in one mole of a compound in a gaseous state is called bond energy. This unit is part of the chemistry library. Bond energies and bond lengths. As we go across a period we see bond length decreases. Bond length and bond strength. A bond is not static but dynamic with the atoms undergoing the attractive and repulsive forces as described in the potential well (fig. Web below is a table of average bond lengths. Browse videos, articles, and exercises by topic. The length of the bond is determined by the number of bonded electrons (the bond order). Browse videos, articles, and exercises by topic. Single and multiple covalent bonds. It is a transferable property of a bond between atoms of fixed types, relatively independent of the rest of the molecule. Web from this graph, we can determine the equilibrium bond length (the internuclear distance at the potential energy minimum) and the bond energy (the energy required to. This unit is part of the chemistry library. Bond length and bond strength. Before we go into the details explaining the bong lengths and bond strengths in organic chemistry, let’s put a small summary for these two properties right from the beginning as it stays relevant for all types of bonds we are going to talk about. This is because. Bond length and bond strength. A bond is not static but dynamic with the atoms undergoing the attractive and repulsive forces as described in the potential well (fig. Single bonds have a bond order of one, and multiple bonds with bond orders of two (a double bond) and three (a triple bond). Web from this graph, we can determine the equilibrium bond length (the internuclear distance at the potential energy minimum) and the bond energy (the energy required to separate the two atoms). As we go across a period we see bond length decreases. Bond order is the number of electron pairs that hold two atoms together. The higher the bond order, the stronger the pull between the two atoms and the shorter the bond length. This is because as we go down the group, the atomic radius increases. This unit is part of the chemistry library. Single and multiple covalent bonds. 8.9.1), where the bottom of the well represents the equilibrium position of the oscillating atoms, which. Web in molecular geometry, bond length or bond distance is defined as the average distance between nuclei of two bonded atoms in a molecule. Before we go into the details explaining the bong lengths and bond strengths in organic chemistry, let’s put a small summary for these two properties right from the beginning as it stays relevant for all types of bonds we are going to talk about. Predicting bond type (metals vs. Bond energies and bond lengths. Browse videos, articles, and exercises by topic.Bond Length and Bond Strength Pathways to Chemistry
PPT Chemical Bonding and Molecular Structure (Chapter 9) PowerPoint
Bond lengths [Å] and angles [°] for 4c Download Scientific Diagram
Selected bond lengths (Å), bond angles ( • ), and intermolecular
PPT 1. Structure and Bonding PowerPoint Presentation, free download
Bond Length and Bond Strength Chemistry Steps
PPT Covalent Bonding PowerPoint Presentation, free download ID1102596
Bond Length and Bond Strength Chemistry Steps
Selected bond lengths (Å) and bond angles (°) for ligand Download Table
Bond Length and Bond Energy
The Length Of The Bond Is Determined By The Number Of Bonded Electrons (The Bond Order).
Web The Amount Of Energy Required To Break All Covalent Bonds Of The Same Type In One Mole Of A Compound In A Gaseous State Is Called Bond Energy.
Web Below Is A Table Of Average Bond Lengths.
Web Bond Length Is Defined As The Distance Between The Centers Of Two Covalently Bonded Atoms.
Related Post:

![Bond lengths [Å] and angles [°] for 4c Download Scientific Diagram](https://www.researchgate.net/publication/333256073/figure/download/tbl2/AS:761180935159811@1558491085331/Bond-lengths-A-and-angles-for-4c.png)





.PNG)