Chemical Nomenclature Chart
Chemical Nomenclature Chart - By the end of this section, you will be able to: Chemical nomenclature is the term given to the naming of compounds. Long before chemists knew the formulas for chemical compounds, they developed a system of nomenclature that gave each compound a unique name. The rules we use depends on the type of compound we are attempting to name. To summarize, many compounds may be classified into five different categories, each of which uses a slightly different, but related, set of rules. Derive names for common types of inorganic compounds using a systematic approach. Web tutorials and problem sets. Different rules apply to each. In this section, we discuss. Web chemical nomenclature | introduction to chemistry. Chemists use specific rules and conventions to name different compounds. These compilations are coordinated by the division of chemical nomenclature and. Each chemical name should refer to a single substance. If only one atom of a specific type is present, no subscript is used. Today we often use chemical formulas, such as nacl, c 12 h 22 o 11, and co (nh 3) 6 (clo 4) 3 , to describe chemical compounds. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. Names that are arbitrary (including the names of the elements, such as sodium and hydrogen) as well as laboratory shorthand names (such as diphos and lithal) are For naming purposes, a chemical compound is treated as a combination of a parent compound (section 5) and characteristic (functional) groups, one of which is The goal of the system is to provide each organic compound with a unique and unambiguous name based on its chemical formula and structure. Web the international union of pure and applied chemistry (iupac) is an international federation of organizations that represents chemists in individual countries. This webpage provides clear examples and explanations for naming different types of molecules and ions, as well as links to other useful resources. This section is designed to help you review some of those rules and conventions. You can choose binary, polyatomic, and variable charge ionic compounds, as well as molecular compounds. Chemists use specific rules and conventions to name. The goal of the system is to provide each organic compound with a unique and unambiguous name based on its chemical formula and structure. Web the system used for naming chemical substances depends on the nature of the molecular units making up the compound. Today we often use chemical formulas, such as nacl, c 12 h 22 o 11, and. Binary compound (metal/nonmetal) with fixed charge cation. Web learn the basic rules and principles of nomenclature for organic and inorganic compounds, according to the iupac standards. For naming purposes, a chemical compound is treated as a combination of a parent compound (section 5) and characteristic (functional) groups, one of which is In this section, we discuss. Derive names for common. These are usually either ions or molecules; These compilations are coordinated by the division of chemical nomenclature and. The rules we use in nomenclature depend on the types of bonds the compound has and these are outlined in figure 2.7.1 2.7. Web the international union of pure and applied chemistry (iupac) is an international federation of organizations that represents chemists. These are usually either ions or molecules; This chapter describes an approach that is used to name simple ionic and molecular compounds, such as [latex]\ce{nacl}[/latex], [latex]\ce{caco3}[/latex], and [latex]\ce{n2o4}[/latex]. Web the primary function of chemical nomenclature is to ensure that a spoken or written chemical name leaves no ambiguity concerning which chemical compound the name refers to: To summarize, many compounds. Web the international union of pure and applied chemistry (iupac) is an international federation of organizations that represents chemists in individual countries. Web the following website provides practice with naming chemical compounds and writing chemical formulas. By the end of this section, you will be able to: Different rules apply to each. For atoms that have two or more present,. These five common classes of compounds are. Web specialists in nomenclature recognise two different categories of nomenclature. You can choose binary, polyatomic, and variable charge ionic compounds, as well as molecular compounds. Each chemical name should refer to a single substance. Web the full text of iupac and iubmb organic and biochemical nomenclature recommendations. Web in this section we will look at nomenclature of simple chemical compounds. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. Different rules apply to each. This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3, and n 2 o 4.. Web nomenclature, a collection of rules for naming things, is important in science and in many other situations. Web the following website provides practice with naming chemical compounds and writing chemical formulas. Long before chemists knew the formulas for chemical compounds, they developed a system of nomenclature that gave each compound a unique name. This module describes an approach that. Different rules apply to each. Each chemical name should refer to a single substance. Today we often use chemical formulas, such as nacl, c 12 h 22 o 11, and co (nh 3) 6 (clo 4) 3 , to describe chemical compounds. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. These. This section is designed to help you review some of those rules and conventions. The rules we use depends on the type of compound we are attempting to name. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. In this section, we discuss. Web specialists in nomenclature recognise two different categories of nomenclature. The nomenclature used most frequently worldwide is the one created and developed by the international union of pure and applied chemistry (iupac). Web in chemical nomenclature, the iupac nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended [1] [2] by the international union of pure and applied chemistry (iupac). These five common classes of compounds are. You can choose binary, polyatomic, and variable charge ionic compounds, as well as molecular compounds. Names that are arbitrary (including the names of the elements, such as sodium and hydrogen) as well as laboratory shorthand names (such as diphos and lithal) are These are usually either ions or molecules; A molecular formula tells us what atoms and how many of each type of atom are present in a molecule. Long before chemists knew the formulas for chemical compounds, they developed a system of nomenclature that gave each compound a unique name. Binary compound (metal/nonmetal) with variable charge cation: Web chemical nomenclature | introduction to chemistry. Binary compound (metal/nonmetal) with fixed charge cation.Chemical Nomenclature and Chemical Formulas Owlcation
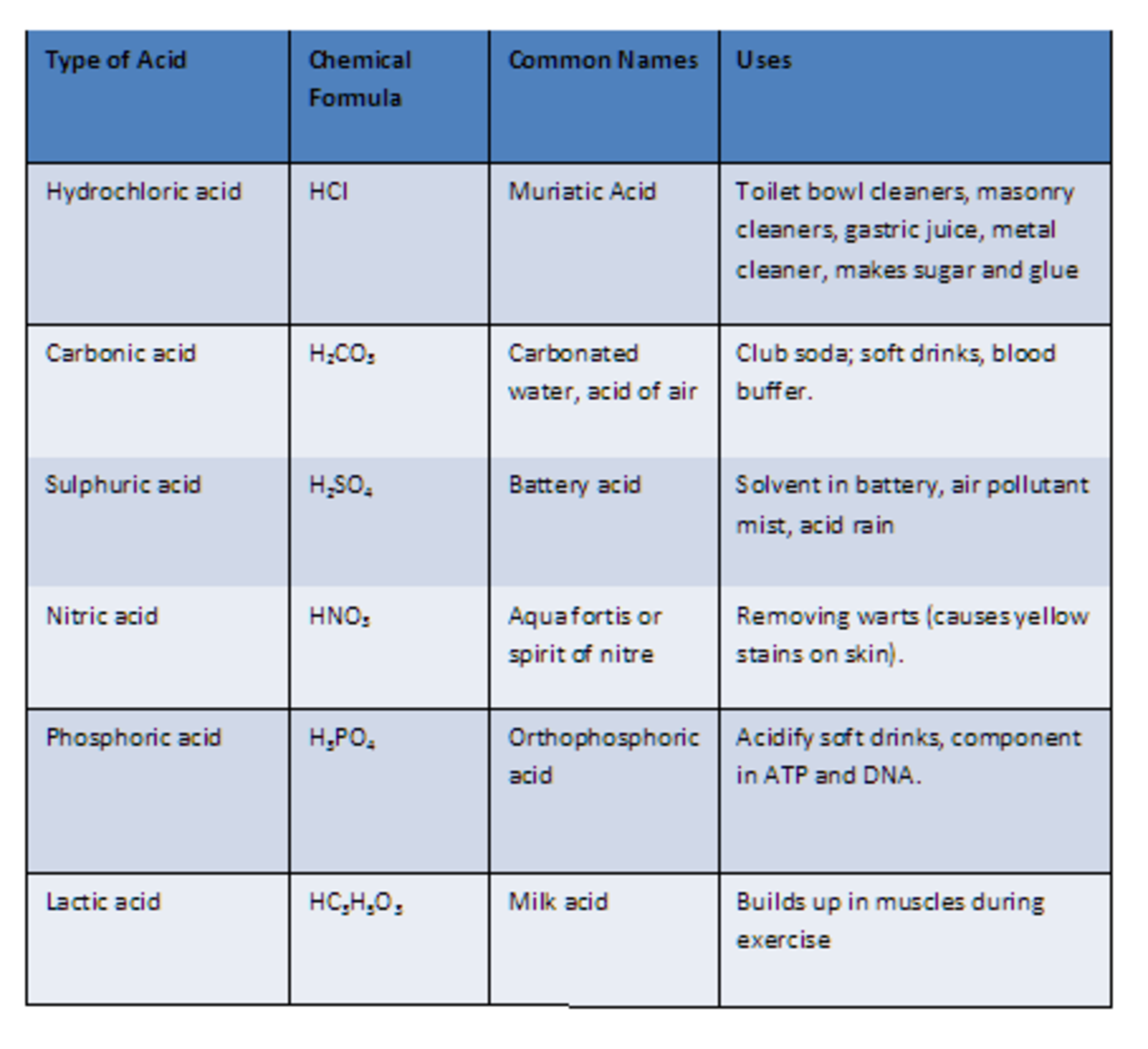
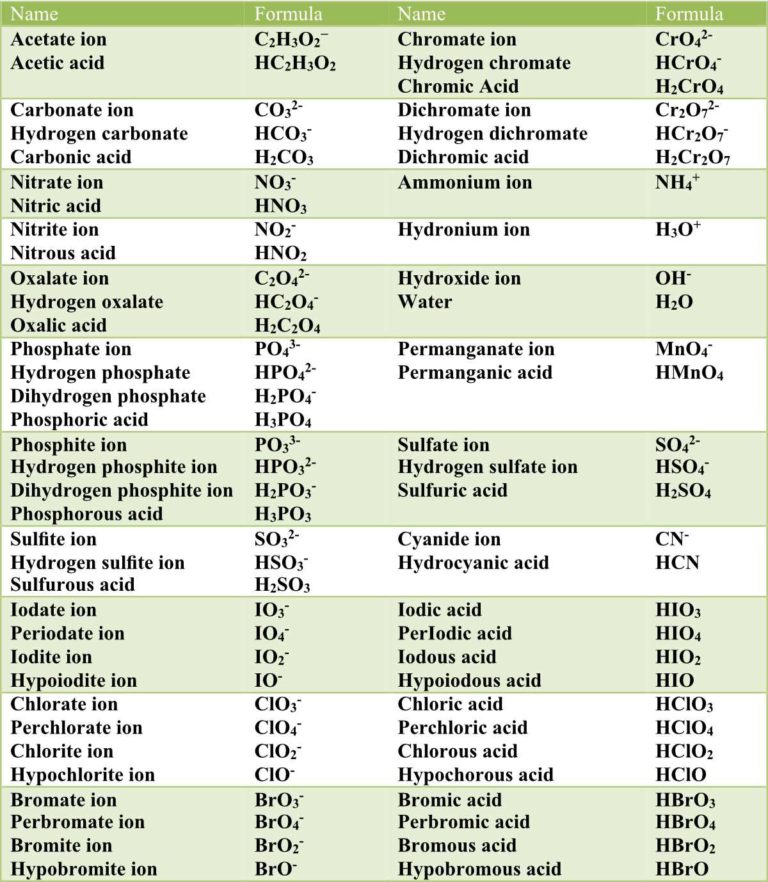
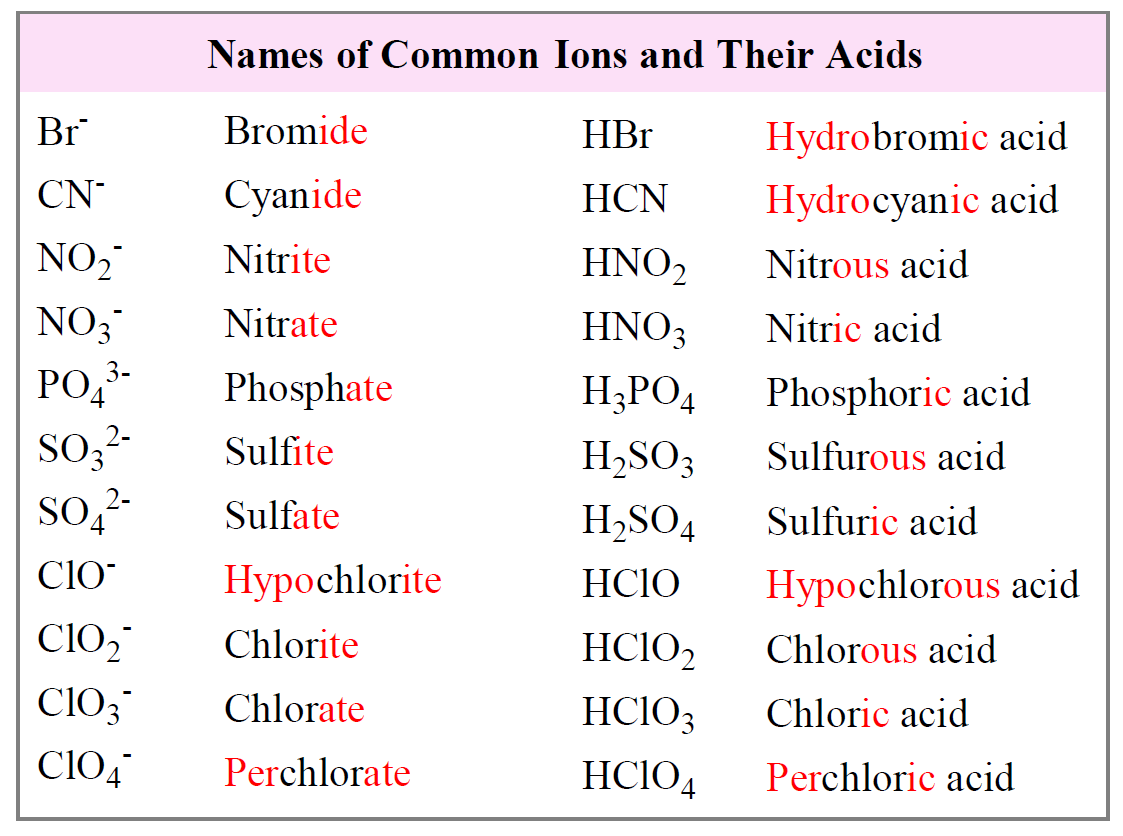
Nomenclature of Acids Pathways to Chemistry
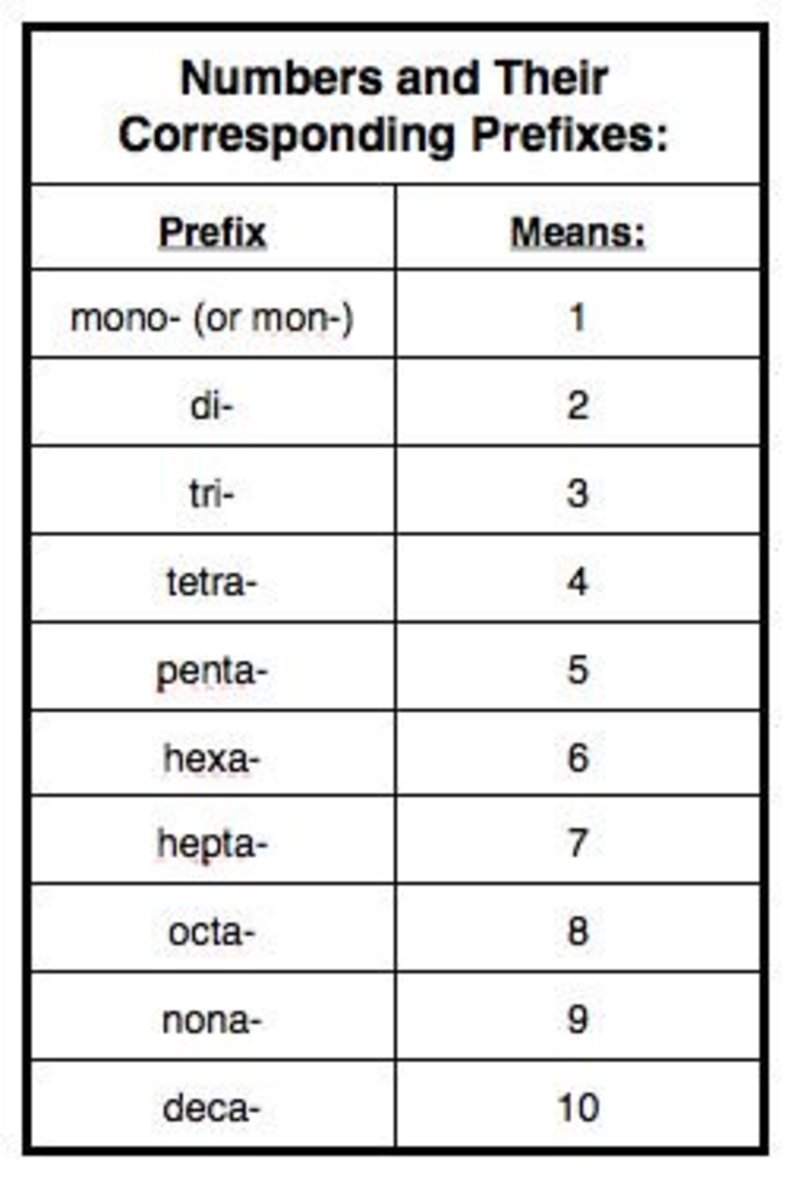
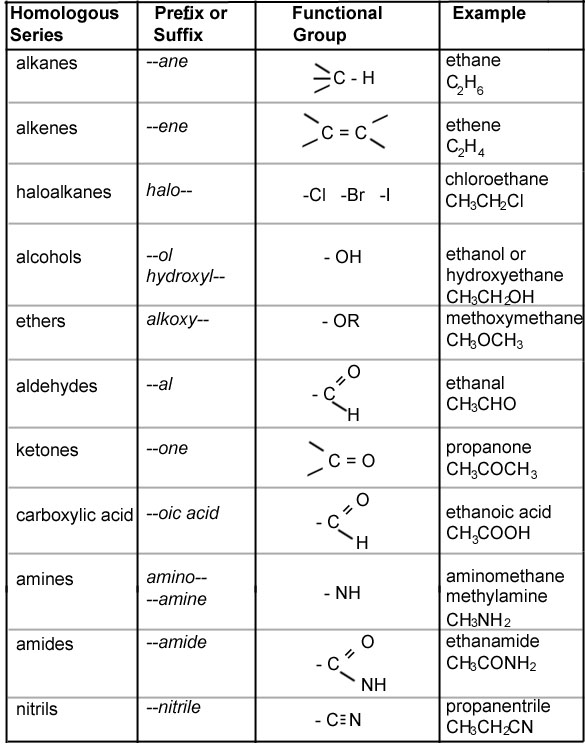
Organic Chemistry Nomenclature Chart
Chemistry Nomenclature Chart
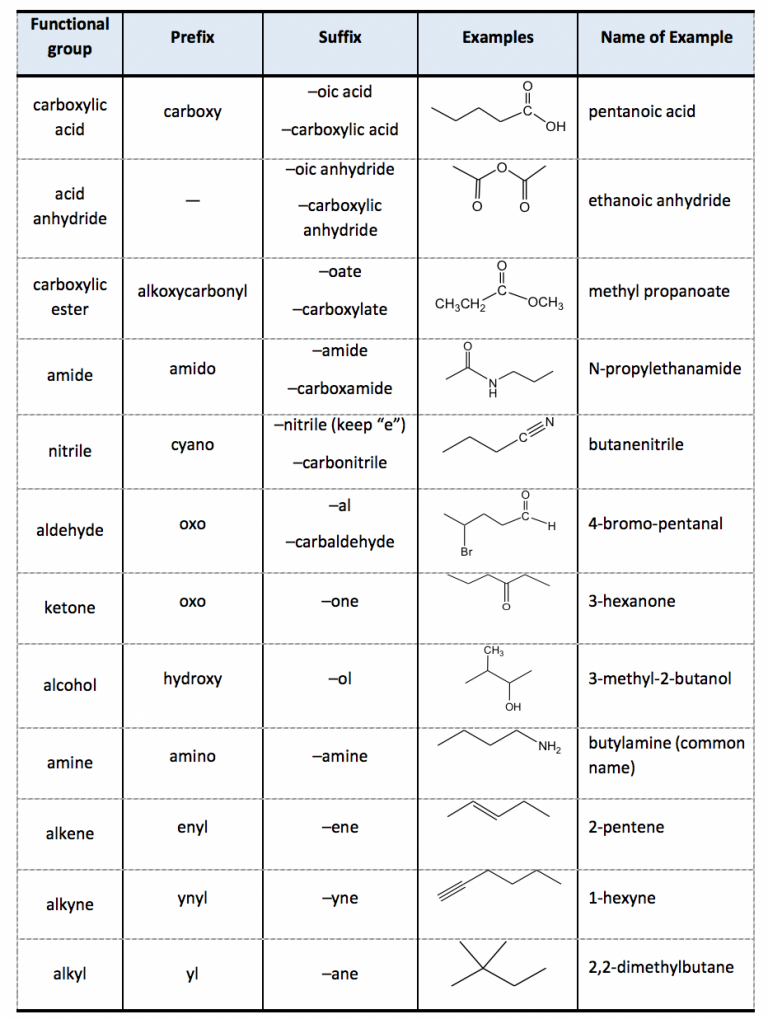
CPSC Organic Chemistry Nomenclature
Naming Acids and Bases Chemistry Steps
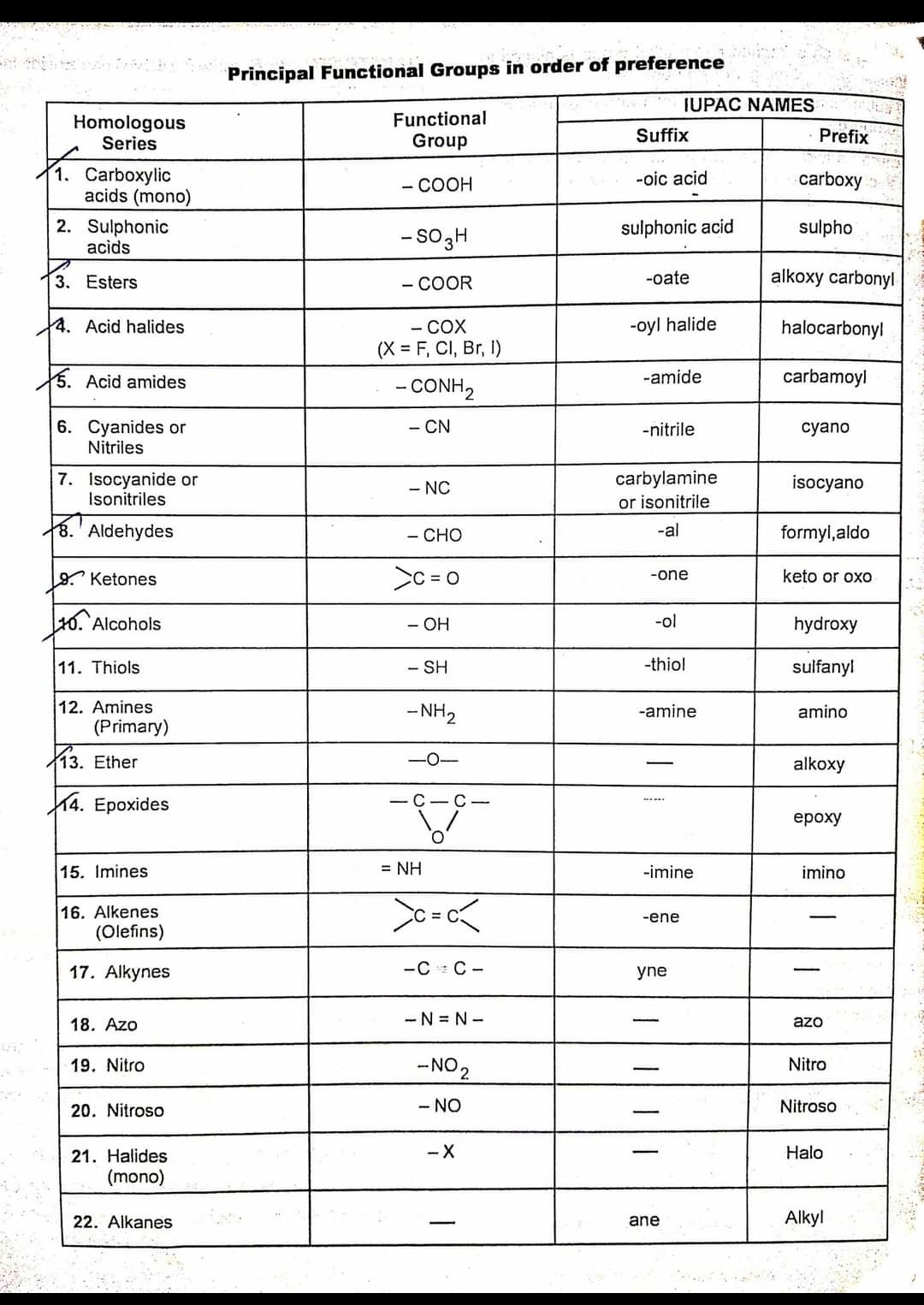
IUPAC Chart Functional Group And Suffix Prefix And Their Principal
Chemistry Nomenclature Chart
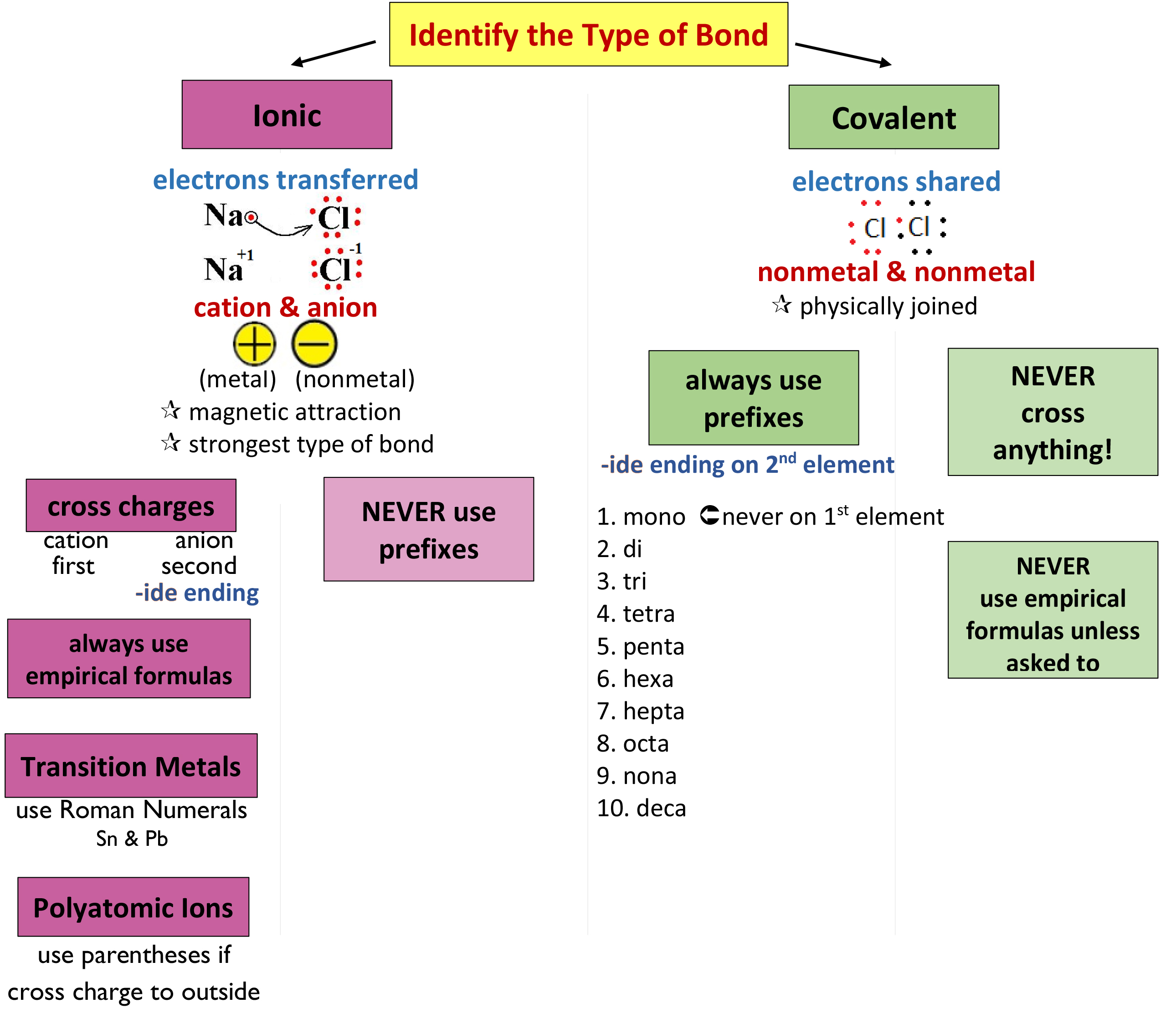
07 Nomenclature Mrs. Cook's Chemistry Class
Chemistry Nomenclature Chart
Chemical Nomenclature Is The Term Given To The Naming Of Compounds.
These Compilations Are Coordinated By The Division Of Chemical Nomenclature And.
Web Iupac Nomenclature Is Used For The Naming Of Chemical Compounds, Based On Their Chemical Composition And Their Structure.
Web Tutorials And Problem Sets.
Related Post: