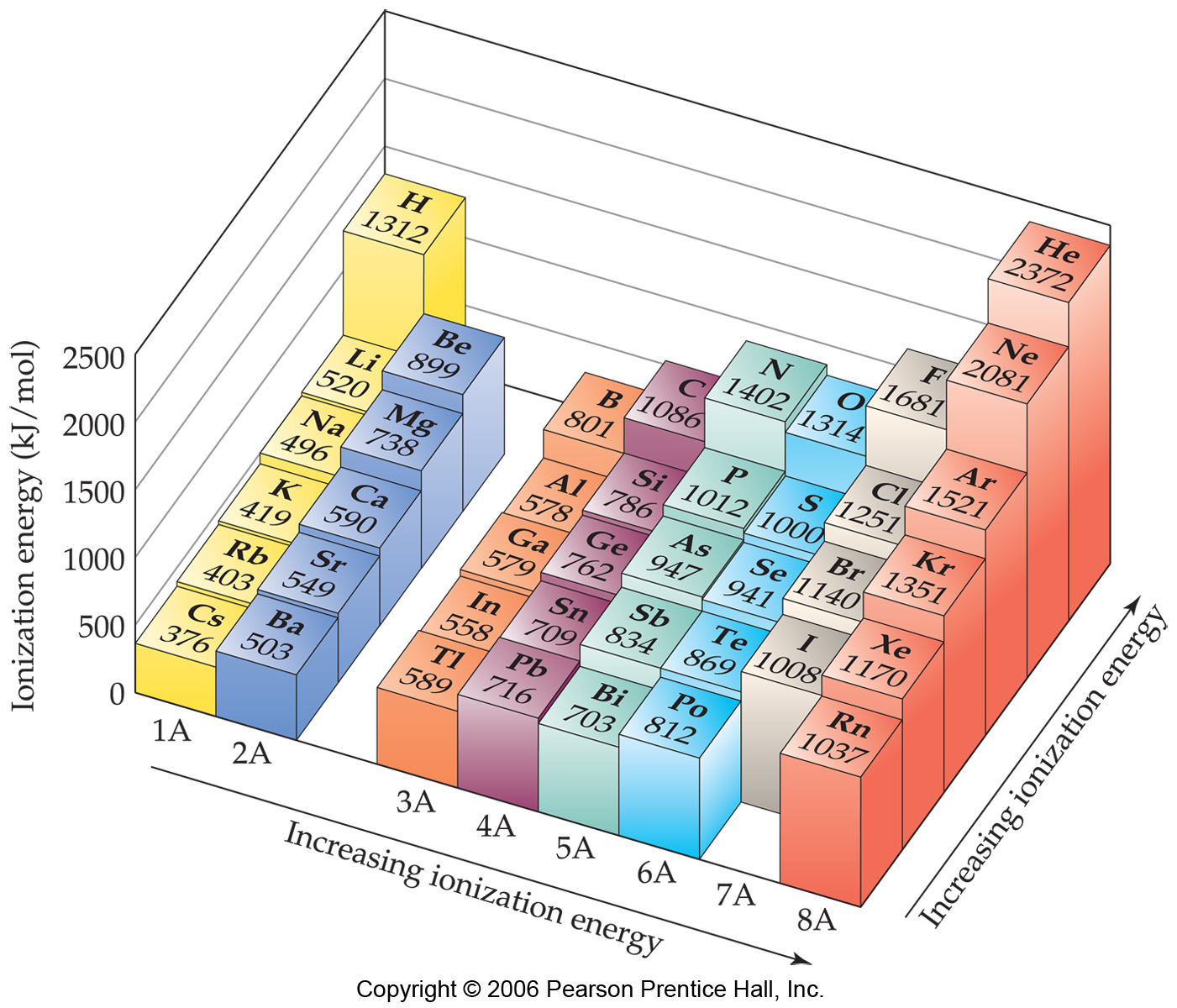
Ionisation Energy Chart
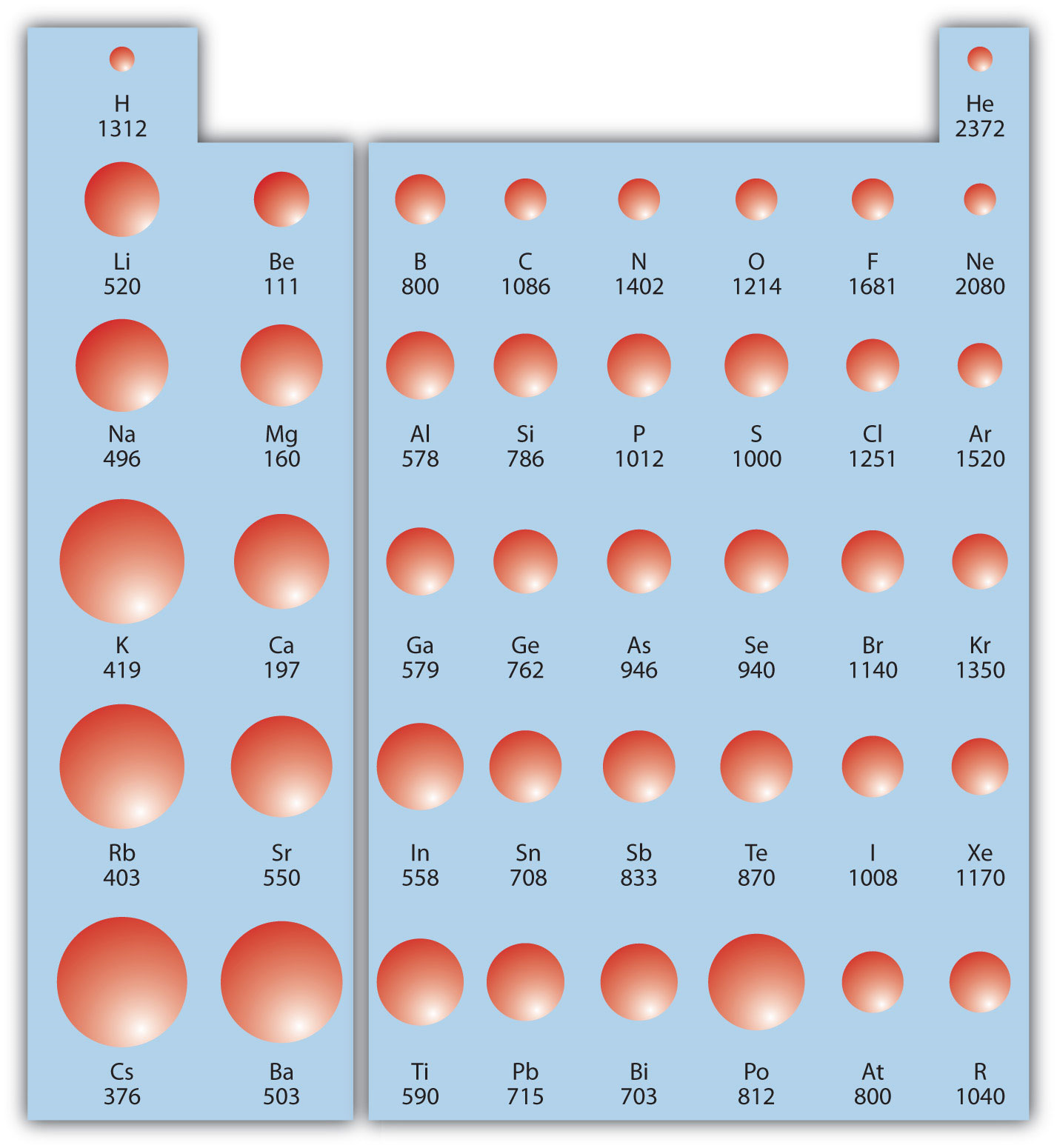
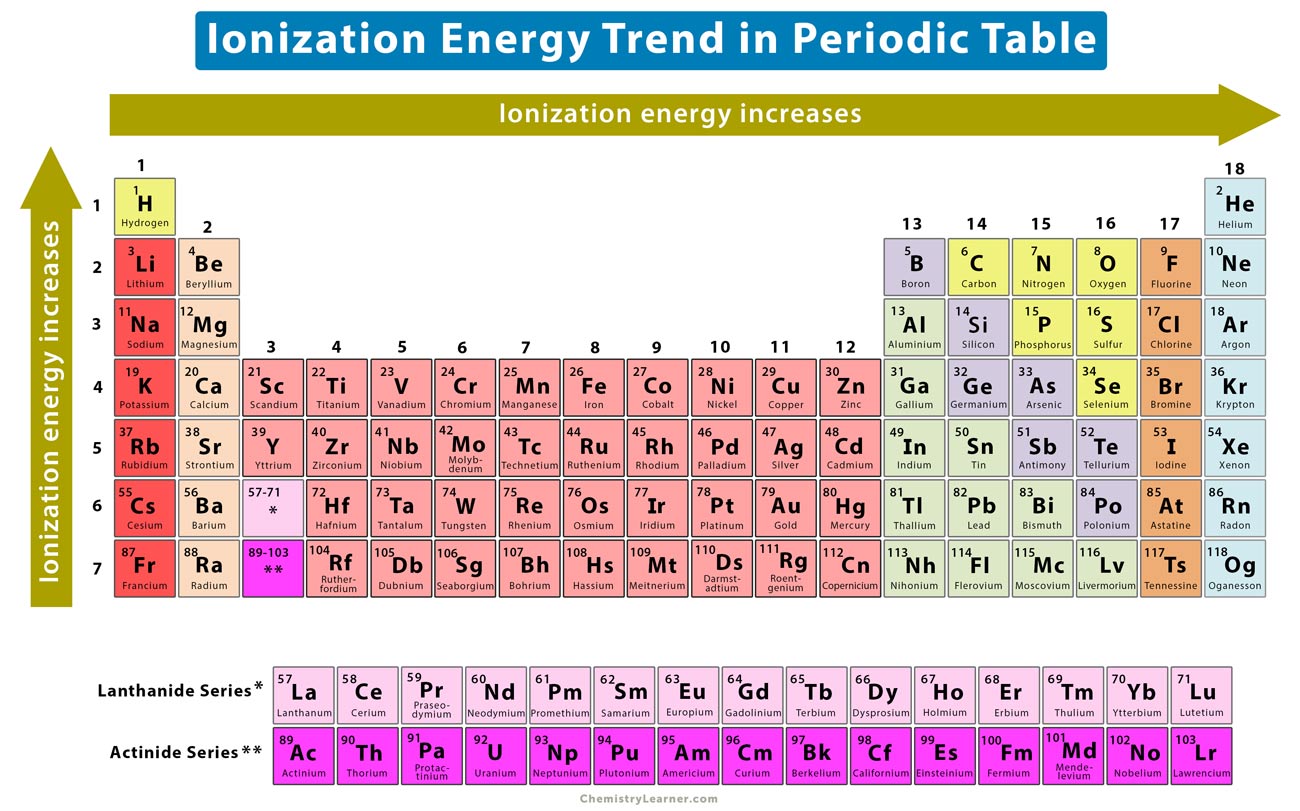
Ionisation Energy Chart - Check out this video for more details: Another is when each of 3 p orbitals have one electron they start to pair as new ones are added (like when moving from nitrogen to oxygen). Web periodic table and trend of ionization energies. Web the 1st ionization energy of the element m is a measure of the energy required to remove one electron from one mole of the gaseous atoms m. The first molar ionization energy applies to the neutral atoms. Web an element's first ionization energy is the energy required to remove the outermost, or least bound, electron from a neutral atom of the element. A high value of ionization energy shows a high attraction between the electron and the nucleus. Learn the definition, trend on the periodic table, first & second ionization energies, see a chart and much more. The energy required to remove an electron is the ionization energy. Image showing periodicity of the chemical elements for ionization energy: Web ionization energy is the energy required to remove an electron from a neutral atom in its gaseous phase. Up to date, curated data provided by mathematica 's elementdata function from wolfram research, inc. Since going from right to left on the periodic table, the atomic radius increases, and the ionization energy increases from left to right in the periods and up the groups. The ionization energy differs for each atom. The table lists only the first ie in ev units. Web values from crc are ionization energies given in the unit ev; On the periodic table, first ionization energy generally increases as you move left to right across a period. Another is when each of 3 p orbitals have one electron they start to pair as new ones are added (like when moving from nitrogen to oxygen). Other values are molar ionization energies given in the unit kj/mol. Check out this video for more details: Image showing periodicity of the chemical elements for ionization energy: Web ionization energy is the amount of energy needed to remove an electron from a neutral gaseous atom and form an ion. Web ionization energy is the energy required to remove an electron from a neutral atom in its gaseous phase. Web values from crc are ionization energies given in. This is the energy per mole necessary to remove electrons from gaseous atoms or atomic ions. Learn its chemical equation, values, trends across a period & down a group, & exception. To convert to kj/mol, multiply by 96.4869. \(i\) is therefore the energy required for the reaction The ionization energy of the elements within a period. Web chemists define the ionization energy (\(i\)) of an element as the amount of energy needed to remove an electron from the gaseous atom \(e\) in its ground state. First ionization energy, second ionization energy as well as third ionization energy of the elements are given in this chart. Web one is that when electrons start to fill p orbital. Web one is that when electrons start to fill p orbital the ionization energy goes down a little. On the periodic table, first ionization energy generally increases as you move left to right across a period. Also, learn first & second ionization energies. Check out this video for more details: Web an element's first ionization energy is the energy required. The stronger an electron is bound to an atom the more ionization energy it requires, therefore these two are directly proportional. Check out this video for more details: The energy required to remove an electron is the ionization energy. Another is when each of 3 p orbitals have one electron they start to pair as new ones are added (like. Up to date, curated data provided by mathematica 's elementdata function from wolfram research, inc. Web explore how ionization energy changes with atomic number in the periodic table of elements via interactive plots. Web values from crc are ionization energies given in the unit ev; \(i\) is therefore the energy required for the reaction Ionization energy is always positive. Click here to buy a book,. Web what is ionization energy. Learn the definition, trend on the periodic table, first & second ionization energies, see a chart and much more. Web ionization is the process of removing an electron from a neutral atom (or compound). First ionization energy, second ionization energy as well as third ionization energy of the elements. Web chemists define the ionization energy (\(i\)) of an element as the amount of energy needed to remove an electron from the gaseous atom \(e\) in its ground state. The energy required to remove an electron is the ionization energy. Web ionization energy is the amount of energy needed to remove an electron from a neutral gaseous atom and form. Web the ionization energy of atoms, denoted e i, is measured by finding the minimal energy of light quanta or electrons accelerated to a known energy that will kick out the least bound atomic electrons. On the periodic table, first ionization energy generally increases as you move left to right across a period. \(i\) is therefore the energy required for. Web ionization energy is the amount of energy needed to remove an electron from a neutral gaseous atom and form an ion. Web ionization energies of the elements. The ionization energy of the elements within a period. The first molar ionization energy applies to the neutral atoms. Web an element's first ionization energy is the energy required to remove the. Learn the definition, trend on the periodic table, first & second ionization energies, see a chart and much more. First ionization energy, second ionization energy as well as third ionization energy of the elements are given in this chart. 1st in a periodic table cityscape style. Web ionization energy is a measure of the energy needed to pull a particular electron away from the attraction of the nucleus. Check out this video for more details: On the periodic table, first ionization energy generally increases as you move left to right across a period. Web periodic table and trend of ionization energies. Web chemists define the ionization energy (\(i\)) of an element as the amount of energy needed to remove an electron from the gaseous atom \(e\) in its ground state. Web an element's first ionization energy is the energy required to remove the outermost, or least bound, electron from a neutral atom of the element. The first molar ionization energy applies to the neutral atoms. Web these tables list values of molar ionization energies, measured in kj⋅mol −1. Web explore how ionization energy changes with atomic number in the periodic table of elements via interactive plots. Web the ionization energy of atoms, denoted e i, is measured by finding the minimal energy of light quanta or electrons accelerated to a known energy that will kick out the least bound atomic electrons. Web ionization energy (the energy associated with forming a cation) decreases down a group and mostly increases across a period because it is easier to remove an electron from a larger, higher energy orbital. Web ionization energy is the energy required to remove an electron from a neutral atom in its gaseous phase. Up to date, curated data provided by mathematica 's elementdata function from wolfram research, inc.Ionization Energy Chart Periodic Table
The Parts of the Periodic Table
Ionization Enthalpy NEET Lab
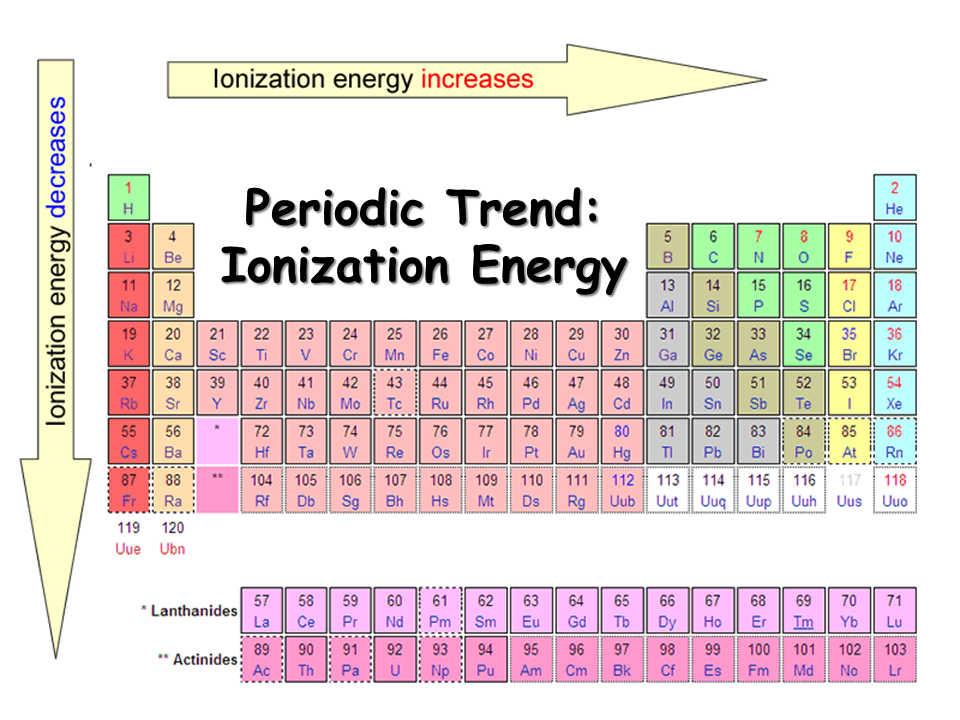
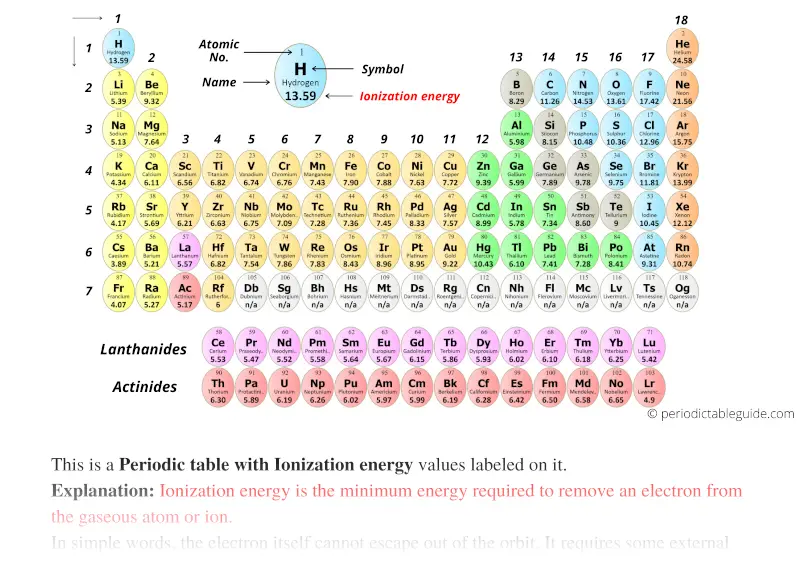
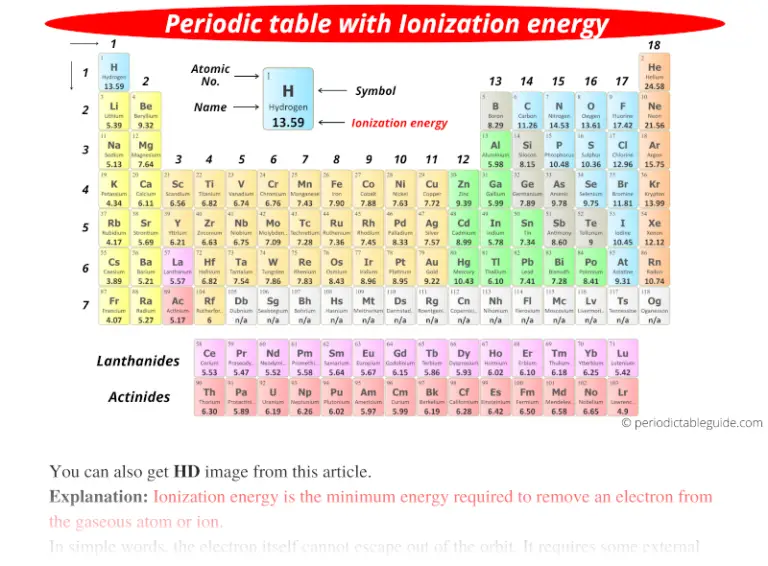
Periodic table with Ionization Energy Values (Labeled Image)
9.9 Periodic Trends Atomic Size, Ionization Energy, and Metallic
Among the Following Which Element Has the Lowest Ionization Energy
Periodic Trends in Ionization Energy CK12 Foundation
Periodic table with Ionization Energy Values (Labeled Image)
8.4 Ionization Energy Chemistry LibreTexts
Pin by Cristina Baldacci on Chemistry Education Chemistry education
\(I\) Is Therefore The Energy Required For The Reaction
Web The 1St Ionization Energy Of The Element M Is A Measure Of The Energy Required To Remove One Electron From One Mole Of The Gaseous Atoms M.
Web First Ionization Energy (Kj/Mol)
Web The Ionization Energy Is A Measure Of The Energy Required To Remove One Electron From One Mole Of Gaseous Atoms Or Ions.
Related Post: