Moles Conversion Chart
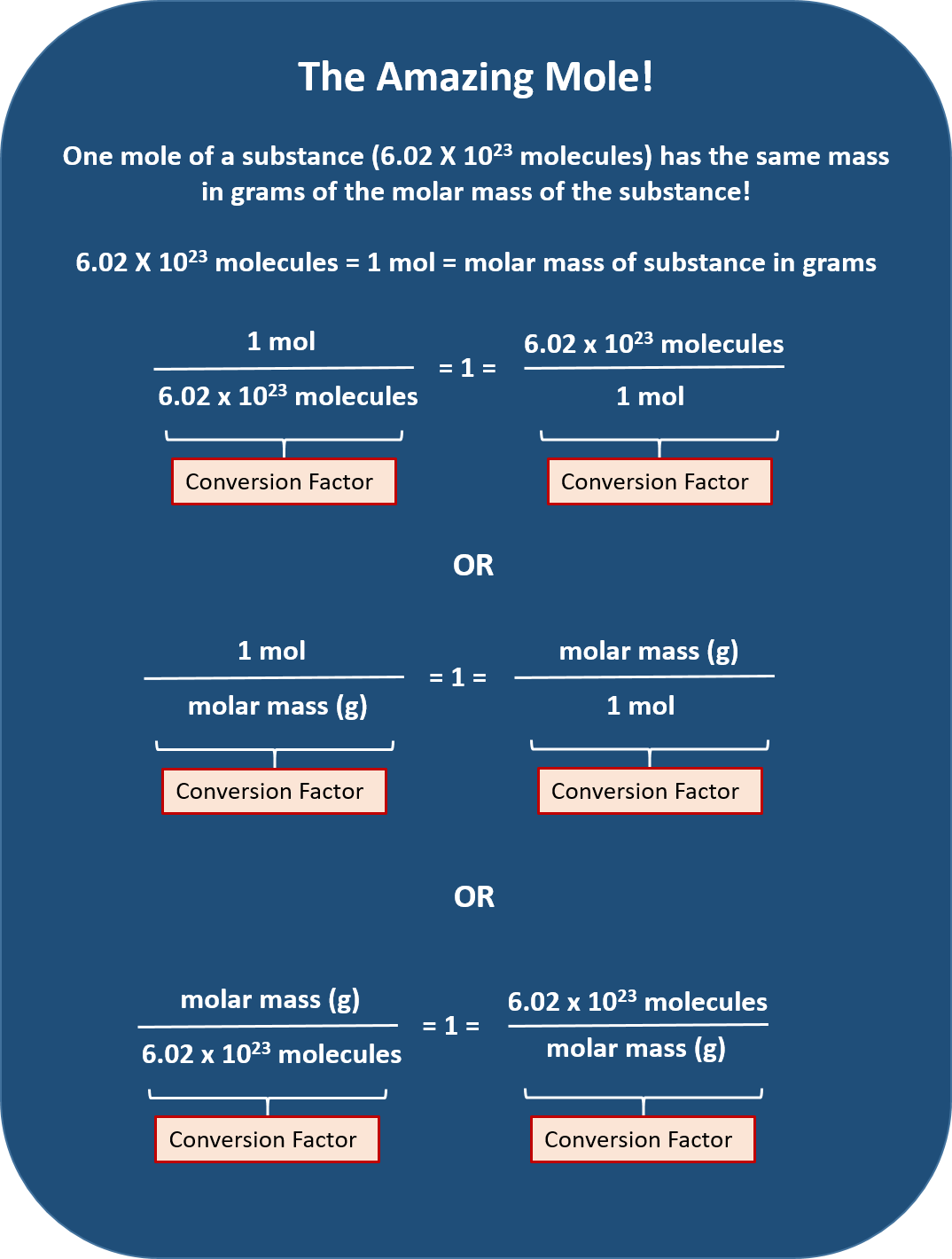
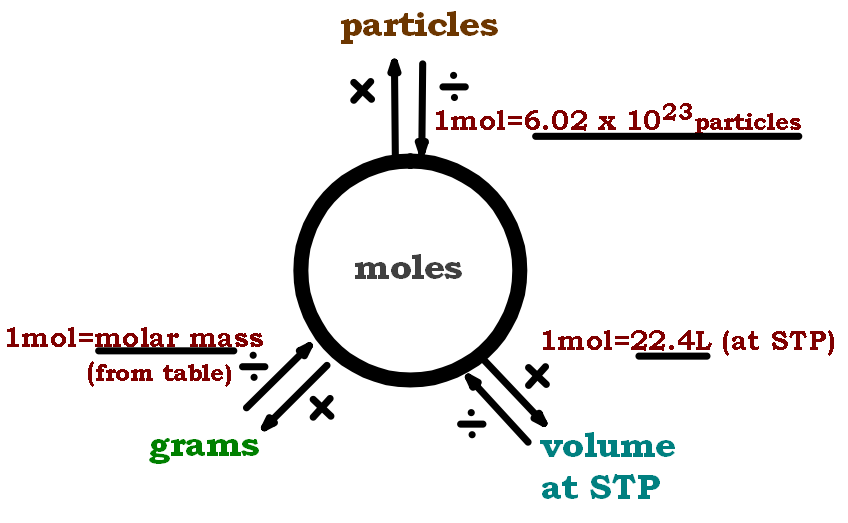
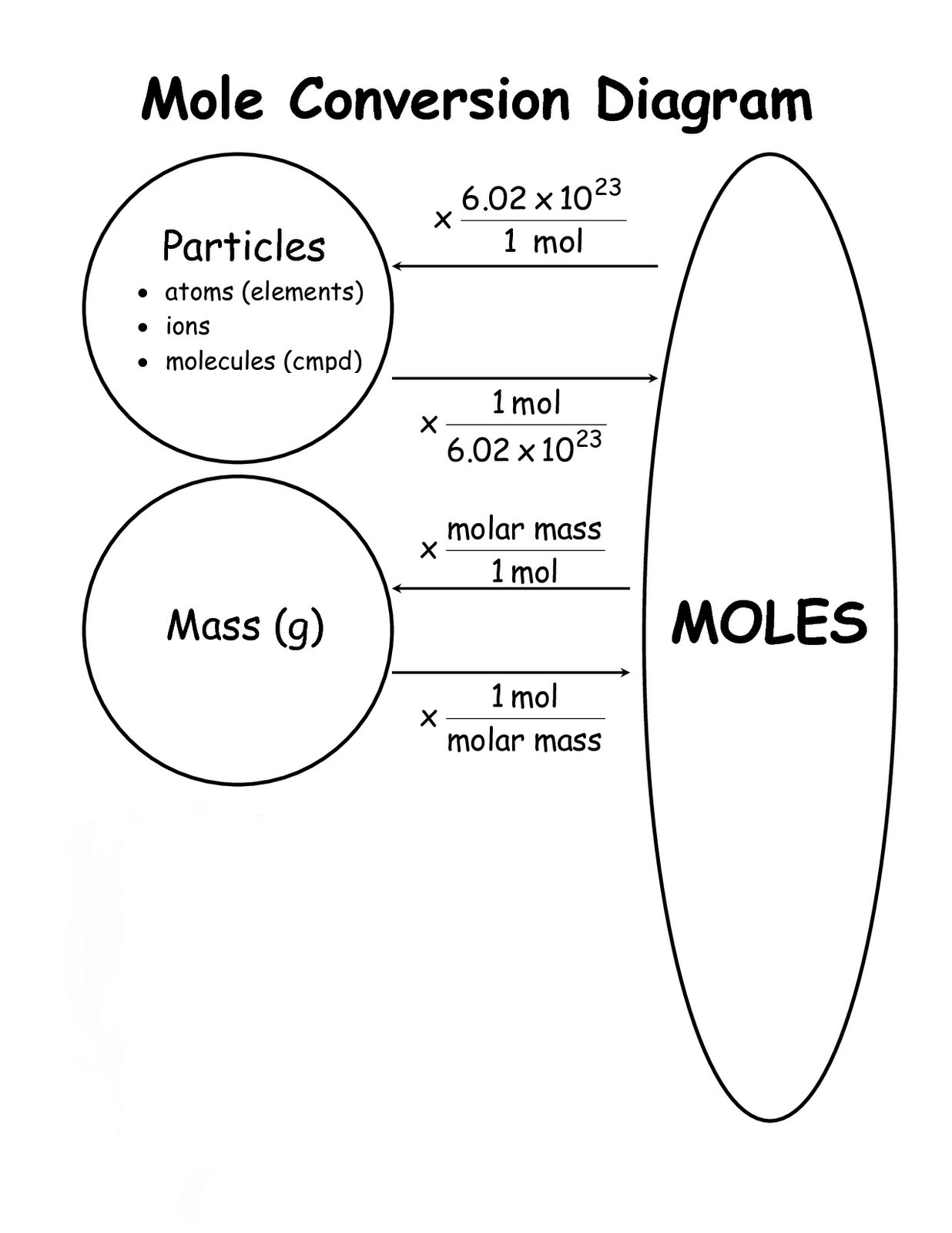
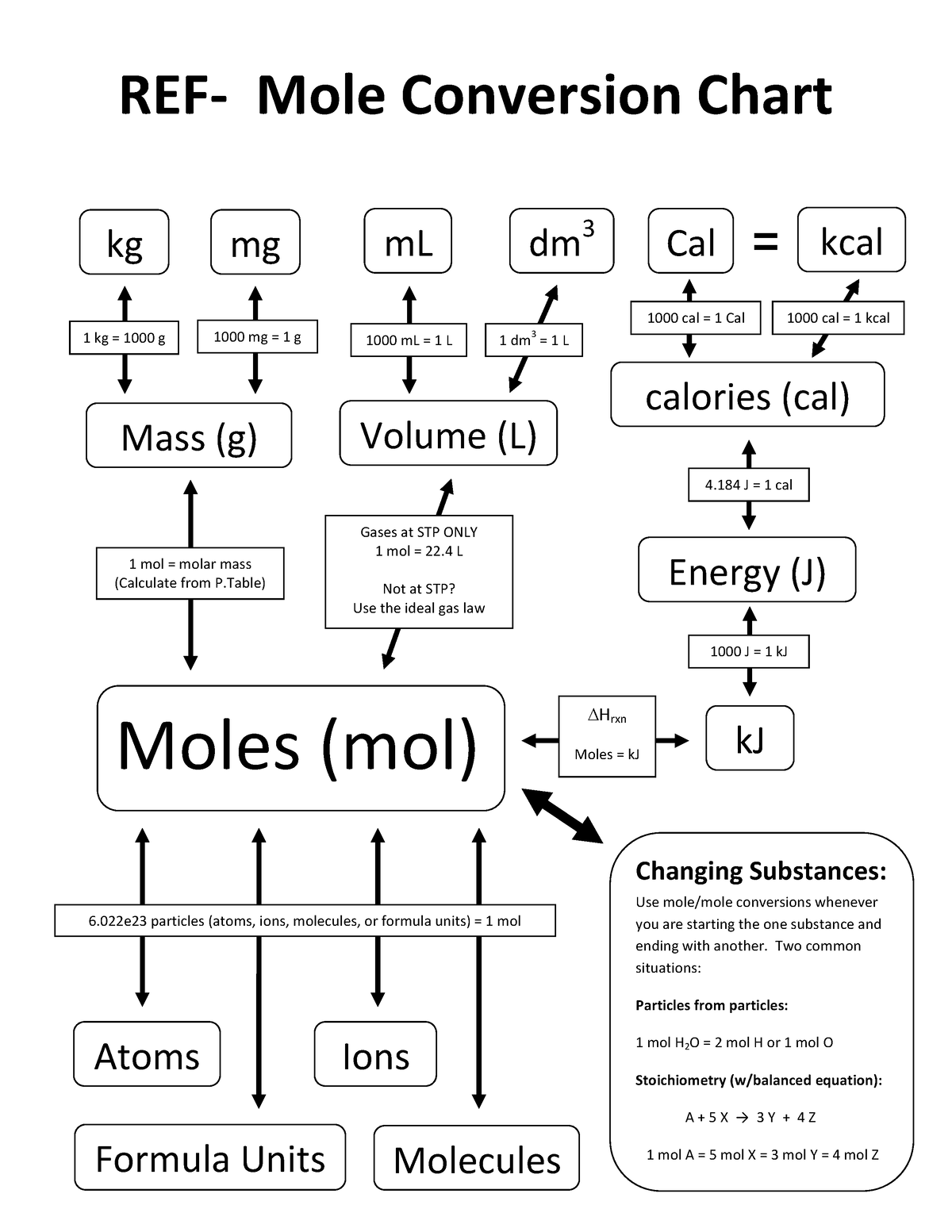
Moles Conversion Chart - Web moles are a type of unit conversion used in chemistry to measure the amount of substances. By converting from moles to grams, liters, and other units, chemists can calculate the mass or volume of a given substance. Web the quantity of substance in moles is equal to the number of molecules divided by the avogadro constant (6.02214076 × 10 23). Divide the mass by the molar mass to find the number of moles in your sample. Web mole conversion chart national mole day october 23. The element carbon exists in. Web whether you’re converting from moles to grams, moles to volume, or moles to particles (atoms or molecules), use this quick guide to remind you of how to do each type of mole conversion: Using our unit conversion techniques, we can use the mole label to convert back and forth between the number of particles and moles. Web to convert from grams to moles, follow these few simple steps: Each set of units must be converted to moles first. Convert from mass or moles of one substance to mass or moles of another substance in a chemical reaction. By converting from moles to grams, liters, and other units, chemists can calculate the mass or volume of a given substance. Web perform conversions between mass and moles of a substance. Divide the mass by the molar mass to find the number of moles in your sample. Find the molar mass of the substance you are analyzing. Using our unit conversion techniques, we can use the mole label to convert back and forth between the number of particles and moles. The study of the numerical relationships between the reactants and the products in balanced chemical reactions is called stoichiometry. Web the mole map is a powerful tool to visualize mole conversions. Measure the mass of your sample in grams. From mass (grams) to moles: Measure the mass of your sample in grams. Web we can use these ratios to determine what amount of a substance, in moles, will react with or produce a given number of moles of a different substance. Web perform conversions between mass and moles of a substance. By converting from moles to grams, liters, and other units, chemists can calculate. Web the mole map is a powerful tool to visualize mole conversions. Web whether you’re converting from moles to grams, moles to volume, or moles to particles (atoms or molecules), use this quick guide to remind you of how to do each type of mole conversion: Use a balanced chemical equation to determine molar. By converting from moles to grams,. Each set of units must be converted to moles first. Use a balanced chemical equation to determine molar. Web conversions between moles and number of particles. Our atoms to moles calculator easily converts atoms or molecules to moles. Measure the mass of your sample in grams. Web a mole road map is a simple guide for converting between units of mass, volume, and number of particles. Use a balanced chemical equation to determine molar. Web the mole map is a powerful tool to visualize mole conversions. Using our unit conversion techniques, we can use the mole label to convert back and forth between the number of. Web mole conversion chart national mole day october 23. The study of the numerical relationships between the reactants and the products in balanced chemical reactions is called stoichiometry. Measure the mass of your sample in grams. From mass (grams) to moles: Web the mole map is a powerful tool to visualize mole conversions. Web whether you’re converting from moles to grams, moles to volume, or moles to particles (atoms or molecules), use this quick guide to remind you of how to do each type of mole conversion: Convert from mass or moles of one substance to mass or moles of another substance in a chemical reaction. One mole is equal to 6.022 x. Using our unit conversion techniques, we can use the mole label to convert back and forth between the number of particles and moles. Web conversions between moles and number of particles. One mole is equal to 6.022 x 10^23 atoms, molecules or other particles. The study of the numerical relationships between the reactants and the products in balanced chemical reactions. Web perform conversions between mass and moles of a substance. Web we can use these ratios to determine what amount of a substance, in moles, will react with or produce a given number of moles of a different substance. One mole is equal to 6.022 x 10^23 atoms, molecules or other particles. Divide your initial mass by the molar mass. The study of the numerical relationships between the reactants and the products in balanced chemical reactions is called stoichiometry. Web mole conversion chart national mole day october 23. Convert from mass or moles of one substance to mass or moles of another substance in a chemical reaction. Divide your initial mass by the molar mass of the compound as determined. Using our unit conversion techniques, we can use the mole label to convert back and forth between the number of particles and moles. Web the mole map is a powerful tool to visualize mole conversions. Web to convert from grams to moles, follow these few simple steps: Find the molar mass of the substance you are analyzing. The study of. Use a balanced chemical equation to determine molar. Web whether you’re converting from moles to grams, moles to volume, or moles to particles (atoms or molecules), use this quick guide to remind you of how to do each type of mole conversion: Web conversions between moles and number of particles. Divide your initial mass by the molar mass of the compound as determined by the periodic table. Using our unit conversion techniques, we can use the mole label to convert back and forth between the number of particles and moles. If you are struggling, it will quickly help you understand how to convert from moles, to grams, liters, molecules, and back. Divide the mass by the molar mass to find the number of moles in your sample. One mole is equal to 6.022 x 10^23 atoms, molecules or other particles. Web perform conversions between mass and moles of a substance. Find the molar mass of the substance you are analyzing. Convert from mass or moles of one substance to mass or moles of another substance in a chemical reaction. The element carbon exists in. From mass (grams) to moles: Web the quantity of substance in moles is equal to the number of molecules divided by the avogadro constant (6.02214076 × 10 23). Web the mole map is a powerful tool to visualize mole conversions. Our atoms to moles calculator easily converts atoms or molecules to moles.PPT Chapter 12 Stoichiometry PowerPoint Presentation, free download
Moles to Grams Conversion Examples
Unit 7 Math of Chemistry Ivy Way Science
Chemistry Mysteries Mole Conversions
Mole Conversion Chart Basicand Advanced Conversions1 REF Mole
Molar Mass Conversion Chart
The Mole Presentation Chemistry
Chapter 6 Quantities in Chemical Reactions Chemistry
Mole Calculation O Level Secondary Chemistry Tuition
PPT Chemistry 20 Mole Conversions PowerPoint Presentation, free
Web Moles Are A Type Of Unit Conversion Used In Chemistry To Measure The Amount Of Substances.
Web To Convert From Grams To Moles, Follow These Few Simple Steps:
Web A Mole Road Map Is A Simple Guide For Converting Between Units Of Mass, Volume, And Number Of Particles.
Measure The Mass Of Your Sample In Grams.
Related Post:



.PNG)