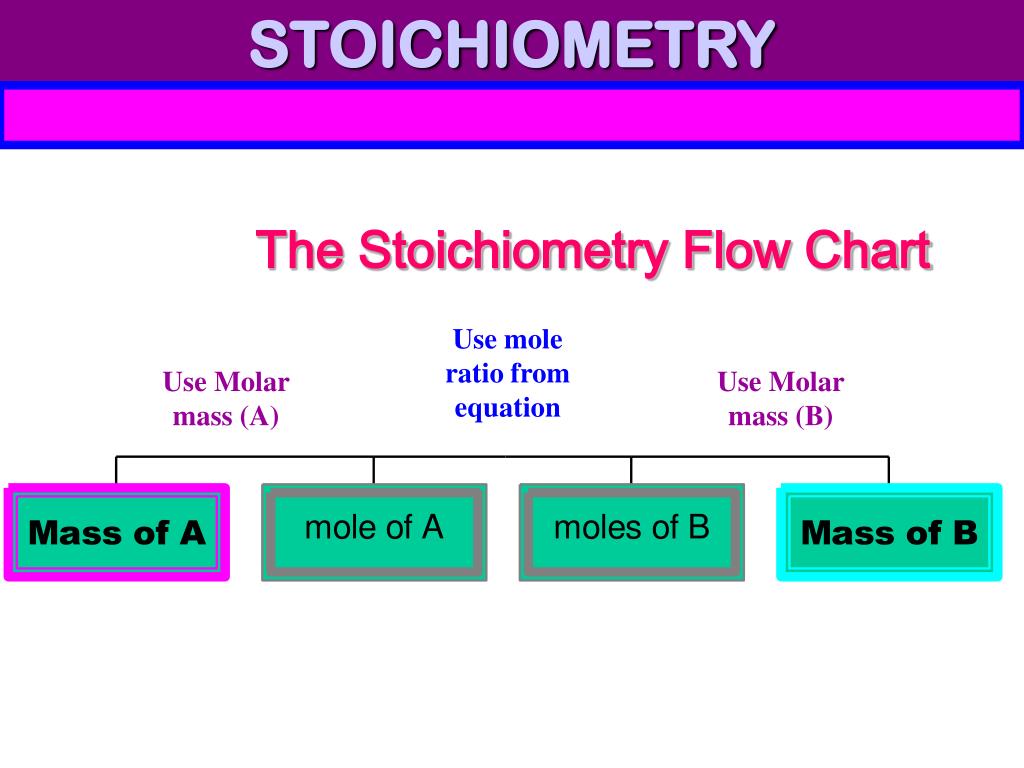
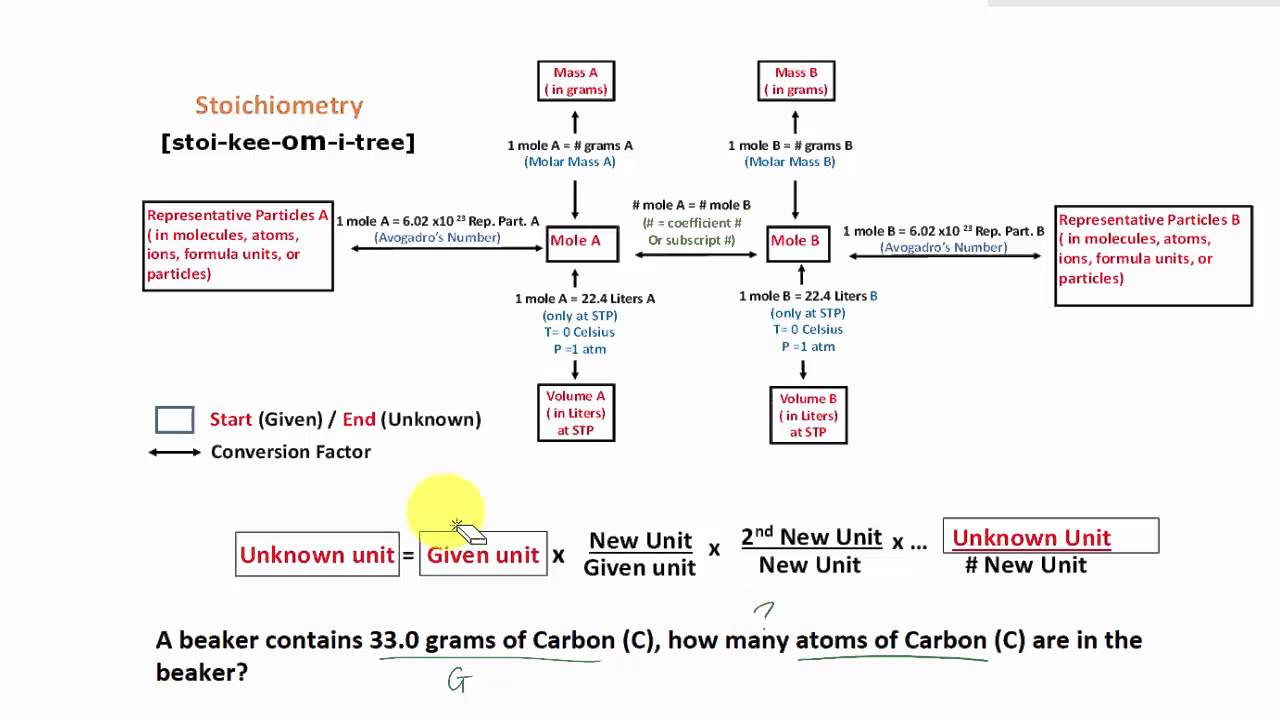
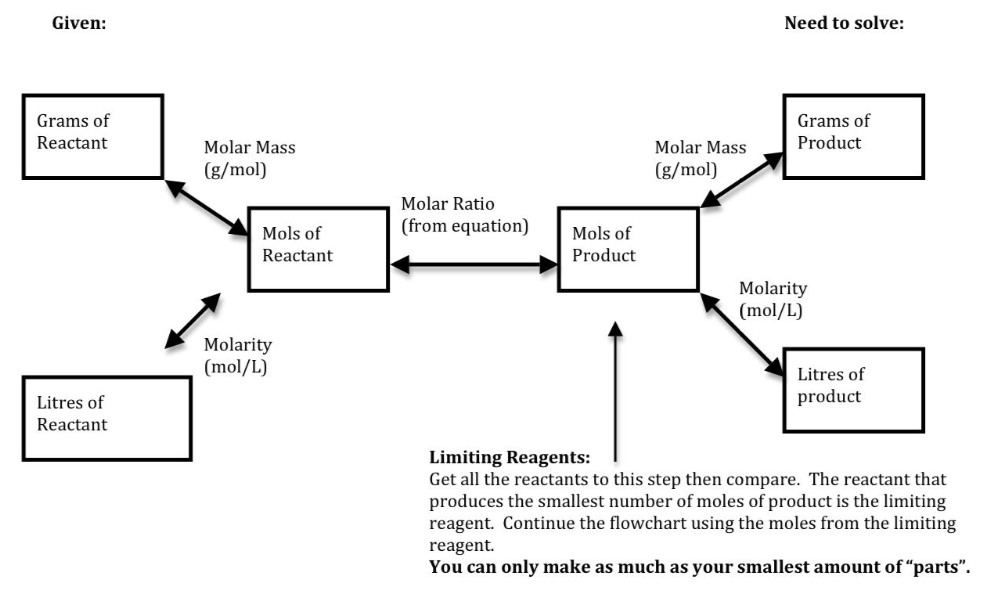
Stoichiometry Flow Chart
Stoichiometry Flow Chart - Moles of a is converted to moles of b by multiplying by the molar ratio. When chemists conduct experiments, they need to know how much of each reactant to use and what amount of product to expect. Web stoichiometry molar mass the trick: Web stoichiometry tutorial for converting mass and moles using stoichiometric conversions, balanced reactions, and molecular weights. Web flow chart for solving stoichiometry problems: Writing and balancing chemical equations; Perform stoichiometric calculations involving mass, moles, and solution molarity. Web a balanced chemical equation shows us the numerical relationships between each of the species involved in the chemical change. Web stoichiometry (/ ˌ s t ɔɪ k i ˈ ɒ m ɪ t r i /) is the relationship between the weights of reactants and products before, during, and following chemical reactions. A stoichiometric quantity of a reactant is the amount necessary to react completely with the other reactant(s). We will start with the simplest types of stoichiometric equations, those involving masses. A stoichiometric quantity of a reactant is the amount necessary to react completely with the other reactant(s). Web explain the concept of stoichiometry as it pertains to chemical reactions. Web derive chemical equations from narrative descriptions of chemical reactions. Web flowchart of steps in stoichiometric calculations. Web stoichiometry is a general term for relationships between amounts of substances in chemical reactions. Web stoichiometry (/ ˌ s t ɔɪ k i ˈ ɒ m ɪ t r i /) is the relationship between the weights of reactants and products before, during, and following chemical reactions. Web explain the concept of stoichiometry as it pertains to chemical reactions; Moles of a is converted to moles of b by multiplying by the molar ratio. Web schematic flowchart showing how density is used for converting volume of pure substance to mass, molar mass for mass and moles, molarity for moles and volume of solution, avogadro's number for moles and number of particles, and stoichiometric factor for relating moles of one substance to another. Web explain the concept of stoichiometry as it pertains to chemical reactions; Web a balanced chemical equation shows us the numerical relationships between each of the species involved in the chemical change. Chose your starting point (“a”) by what units you have to start with: Web flow chart for solving stoichiometry problems: Web stoichiometry (/ ˌ s t ɔɪ k. It also describes calculations done to determine how much of a substance will be used in a reaction, left over after a reaction, produced by a. Web the stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation. Web at its core, stoichiometry is the study of the quantitative relationships between the reactants. When chemists conduct experiments, they need to know how much of each reactant to use and what amount of product to expect. Use balanced chemical equations to derive stoichiometric factors relating amounts of reactants and products; Web explain the concept of stoichiometry as it pertains to chemical reactions; Moles of a is converted to moles of b by multiplying by. Web stoichiometry, flow chart, moles to mass conversion. Coefficients tell the number of molecules or entities. Web figure 1.3.2 provides a general outline of the various computational steps associated with many reaction stoichiometry calculations. We can use these numerical relationships to write mole ratios, which allow us to convert between amounts of reactants and/or products (and thus solve stoichiometry problems!).. Web stoichiometry tutorial for converting mass and moles using stoichiometric conversions, balanced reactions, and molecular weights. The flow chart depicts the various computational steps involved in most reaction stoichiometry calculations. Web derive chemical equations from narrative descriptions of chemical reactions. Web stoichiometry flow chart | creately. Coefficients tell the number of molecules or entities. Perform stoichiometric calculations involving mass, moles, and solution molarity Perform stoichiometric calculations involving mass and moles. Web schematic flowchart showing how density is used for converting volume of pure substance to mass, molar mass for mass and moles, molarity for moles and volume of solution, avogadro's number for moles and number of particles, and stoichiometric factor for relating moles of. Web explain the concept of stoichiometry as it pertains to chemical reactions; Find the empirical formula of paba. Perform stoichiometric calculations involving mass, moles, and solution molarity. Web one can calculate the empirical formula from the percent composition. Web schematic flowchart showing how density is used for converting volume of pure substance to mass, molar mass for mass and moles,. Flow chart showing three steps for using a balanced chemical equation to relate measured quantities of various reactants and products to each other. When chemists conduct experiments, they need to know how much of each reactant to use and what amount of product to expect. Moles of a is converted to moles of b by multiplying by the molar ratio.. Mass (g,kg, volume (l, or with. Flow chart showing three steps for using a balanced chemical equation to relate measured quantities of various reactants and products to each other. Web covers the steps of basic conversions within stoichiometry without using dimensional analysis. The flow chart depicts the various computational steps involved in most reaction stoichiometry calculations. Web a balanced chemical. Mass (g,kg, volume (l, or with. Web derive chemical equations from narrative descriptions of chemical reactions. Web stoichiometry is a general term for relationships between amounts of substances in chemical reactions. Web stoichiometry, flow chart, moles to mass conversion. Use balanced chemical equations to derive stoichiometric factors relating amounts of reactants and products; Writing and balancing chemical equations; Web flow chart for solving stoichiometry problems: Use balanced chemical equations to derive stoichiometric factors relating amounts of reactants and products; You must start with a balanced chemical equation. Web derive chemical equations from narrative descriptions of chemical reactions. Use balanced chemical equations to derive stoichiometric factors relating amounts of reactants and products. You can easily edit this template using creately. Coefficients tell the number of molecules or entities. Moles of a is converted to moles of b by multiplying by the molar ratio. Moles of b is converted to grams of b by the molar mass. Web stoichiometry flow chart | creately. The preceding chapter introduced the use of element symbols to represent individual atoms. When chemists conduct experiments, they need to know how much of each reactant to use and what amount of product to expect. Web covers the steps of basic conversions within stoichiometry without using dimensional analysis. Use balanced chemical equations to derive stoichiometric factors relating amounts of reactants and products; We can use these numerical relationships to write mole ratios, which allow us to convert between amounts of reactants and/or products (and thus solve stoichiometry problems!).Stoichiometric Calculations CK12 Foundation
PPT STOICHIOMETRY PowerPoint Presentation, free download ID4499255
Flow chart for Stoichiometry classroom Pinterest
PPT STOICHIOMETRY PowerPoint Presentation, free download ID4499255
Extended Reaction Stoichiometry Road Map — Examples Expii
Stoichiometry Flow Chart YouTube
PPT Chapter 12 Stoichiometry PowerPoint Presentation, free download
stoichiometry flowchart introduction YouTube
Stoichiometry Flow Chart
Stoichiometry CHEMISTRY HELP
A Stoichiometric Quantity Of A Reactant Is The Amount Necessary To React Completely With The Other Reactant(S).
Write And Balance Chemical Equations In Molecular, Total Ionic, And Net Ionic Formats.
The Flow Chart Depicts The Various Computational Steps Involved In Most Reaction Stoichiometry Calculations.
We Will Start With The Simplest Types Of Stoichiometric Equations, Those Involving Masses.
Related Post: